The safety and efficacy of once-daily ARAZLO Lotion were assessed in 2 multicenter, randomized, double-blind phase 3 clinical trials of 1,614 subjects 9 years of age and older with facial acne vulgaris. Enrolled subjects had a score of moderate (3) or severe (4) on the Evaluator’s Global Severity Score (EGSS), 20 to 50 inflammatory lesions (papules, pustules, and nodules), 25 to 100 noninflammatory lesions (open and closed comedones), and 2 or fewer facial nodules. The coprimary efficacy endpoints of success on the EGSS, absolute change in noninflammatory lesion count, and absolute change in inflammatory lesion count were assessed at week 12.3
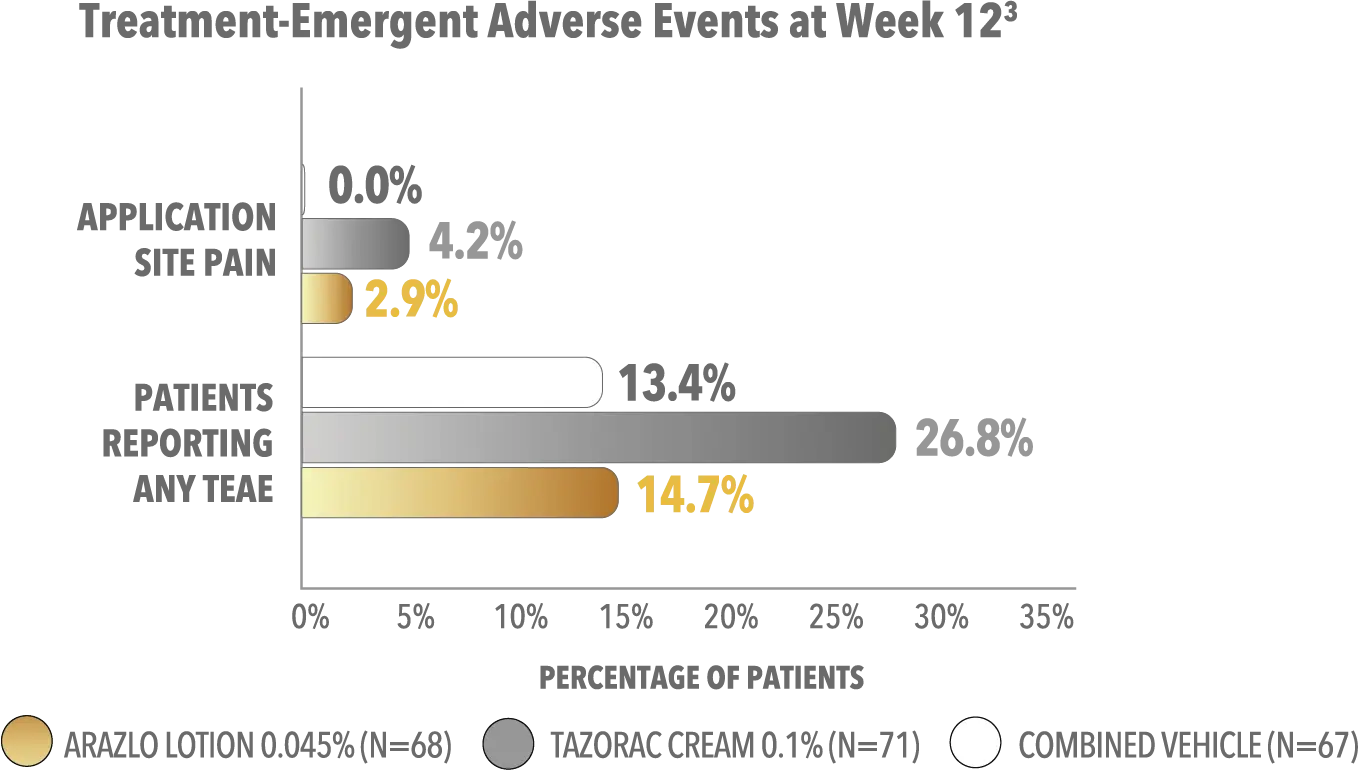
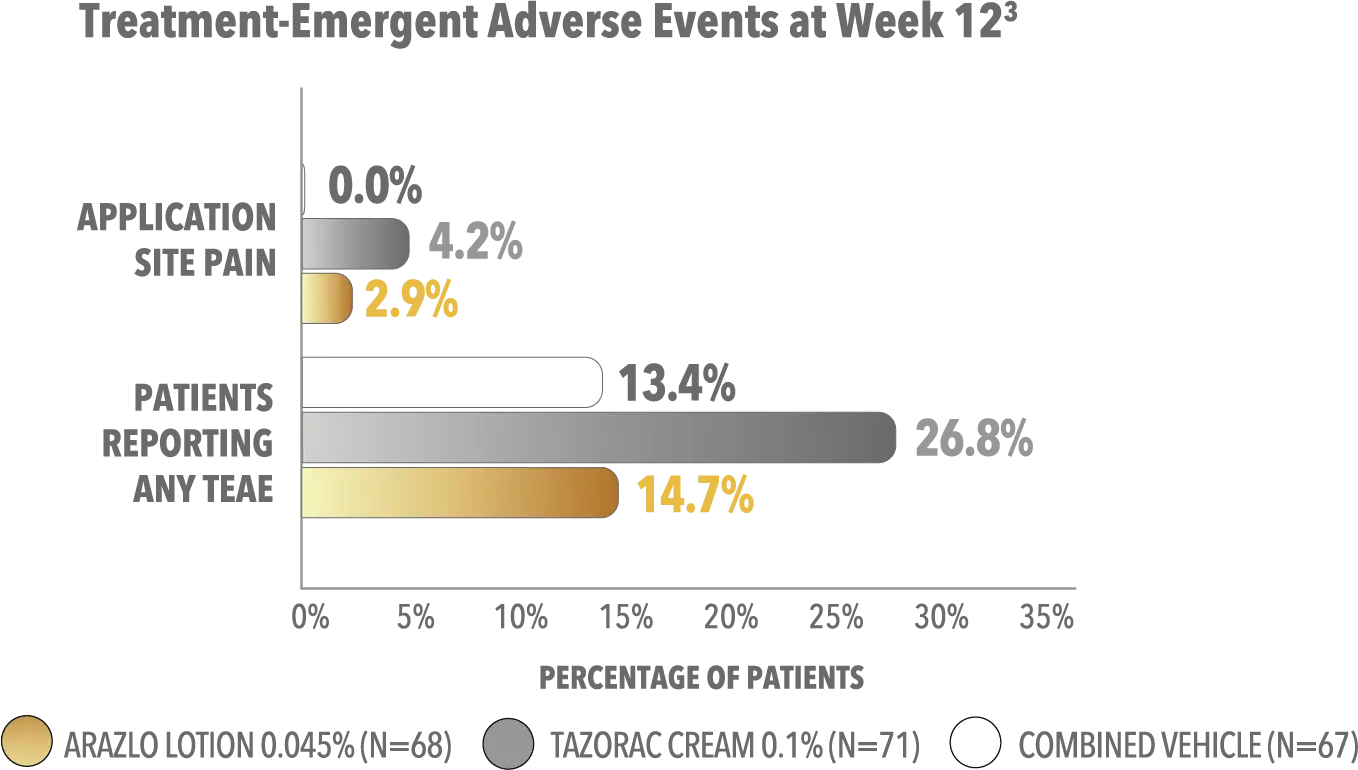
The safety and efficacy of ARAZLO Lotion were assessed in a multicenter, randomized, double-blind, vehicle-controlled phase 2 clinical trial of 210 subjects aged 12 years and older with acne vulgaris. Subjects had a score of moderate (3) or severe (4) on the Evaluator’s Global Severity Score (EGSS), 20 to 40 inflammatory lesions, 20 to 100 noninflammatory lesions, and 2 nodules or less. Subjects were randomized to receive ARAZLO Lotion 0.045%, tazarotene cream 0.1%, or respective vehicle. The week 12 coprimary endpoints included: mean absolute change in inflammatory and noninflammatory lesion counts; and percentage of subjects with at least a 2-grade reduction in EGSS and scores of clear (0) or almost clear (1).1
TEAE, treatment-emergent adverse event.
ARAZLO® (tazarotene) Lotion, 0.045% is indicated for the topical treatment of acne vulgaris in patients 9 years of age and older.
ARAZLO Lotion is for topical use only. Not for oral, ophthalmic, or intravaginal use.
Contraindication
ARAZLO Lotion is contraindicated in pregnancy due to the potential harm to the fetus.
Warnings and Precautions
Embryofetal Risk Females of childbearing potential should be warned of the potential risk and use adequate birth-control measures when ARAZLO Lotion is used. A negative result for pregnancy should be obtained within 2 weeks prior to ARAZLO Lotion therapy. ARAZLO Lotion therapy should begin during a menstrual period.
Skin Irritation Patients using ARAZLO Lotion may experience application site pain, dryness, exfoliation, erythema, and pruritus. Depending upon severity of these adverse reactions, instruct patients to use a moisturizer, reduce the frequency of the application of ARAZLO Lotion, or discontinue use. Therapy can be resumed, or the frequency of application can be increased, as the patient becomes able to tolerate treatment. Avoid application of ARAZLO Lotion to eczematous or sunburned skin.
Ultraviolet Light and Environmental Exposure Minimize unprotected exposure to ultraviolet light, including sunlight and sunlamps, during the use of ARAZLO Lotion. Warn patients who normally experience high levels of sun exposure and those with inherent sensitivity to sun to exercise caution. Use sunscreen products and protective clothing over treated areas when sun exposure cannot be avoided. ARAZLO Lotion should be administered with caution if the patient is also taking drugs known to be photosensitizers (eg, thiazides, tetracyclines, fluoroquinolones, phenothiazines, sulfonamides) because of the increased possibility of augmented photosensitivity. Weather extremes, such as wind or cold, may be more irritating to patients using ARAZLO Lotion.
Adverse Reactions The most common adverse reactions (in ≥1% of patients and greater than vehicle) were: pain, dryness, exfoliation, erythema, and pruritus (all at application site).
To report SUSPECTED ADVERSE REACTIONS, contact Bausch Health US, LLC at 1-800-321-4576 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch. Please click here for full Prescribing Information.
References: 1. ARAZLO Lotion [prescribing information]. Bridgewater, NJ. Bausch Health US, LLC. 2. Tanghetti EA, Kircik LH, Green LJ, et al. A phase 2, multicenter, double-blind, randomized, vehicle-controlled clinical study to compare the safety and efficacy of a novel tazarotene 0.045% lotion and tazarotene 0.1% cream in the treatment of moderate-to-severe acne vulgaris. J Drugs Dermatol. 2019;18(6):542-548. 3. Food and Drug Administration. Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. https://www.accessdata.fda.gov/scripts/cder/ob/index.cfm. Accessed October 10, 2019. 4. Data on file.